
Why Most RWE Fails Payer Scrutiny (and What Actually Passes)
Real-world evidence is no longer optional in market access discussions. Payers expect it. HTA bodies ask for it. Internal teams invest heavily in generating it. And yet, a quiet reality persists: most ...

The Hidden Gap Between Phase III Success and Market Access: When Evidence Misalignment Delays Commercial Readiness
For many biopharma teams, Phase III is treated as the final mountain to climb. Once efficacy and safety are proven, commercial readiness is assumed to follow naturally. In reality, that assumption is ...
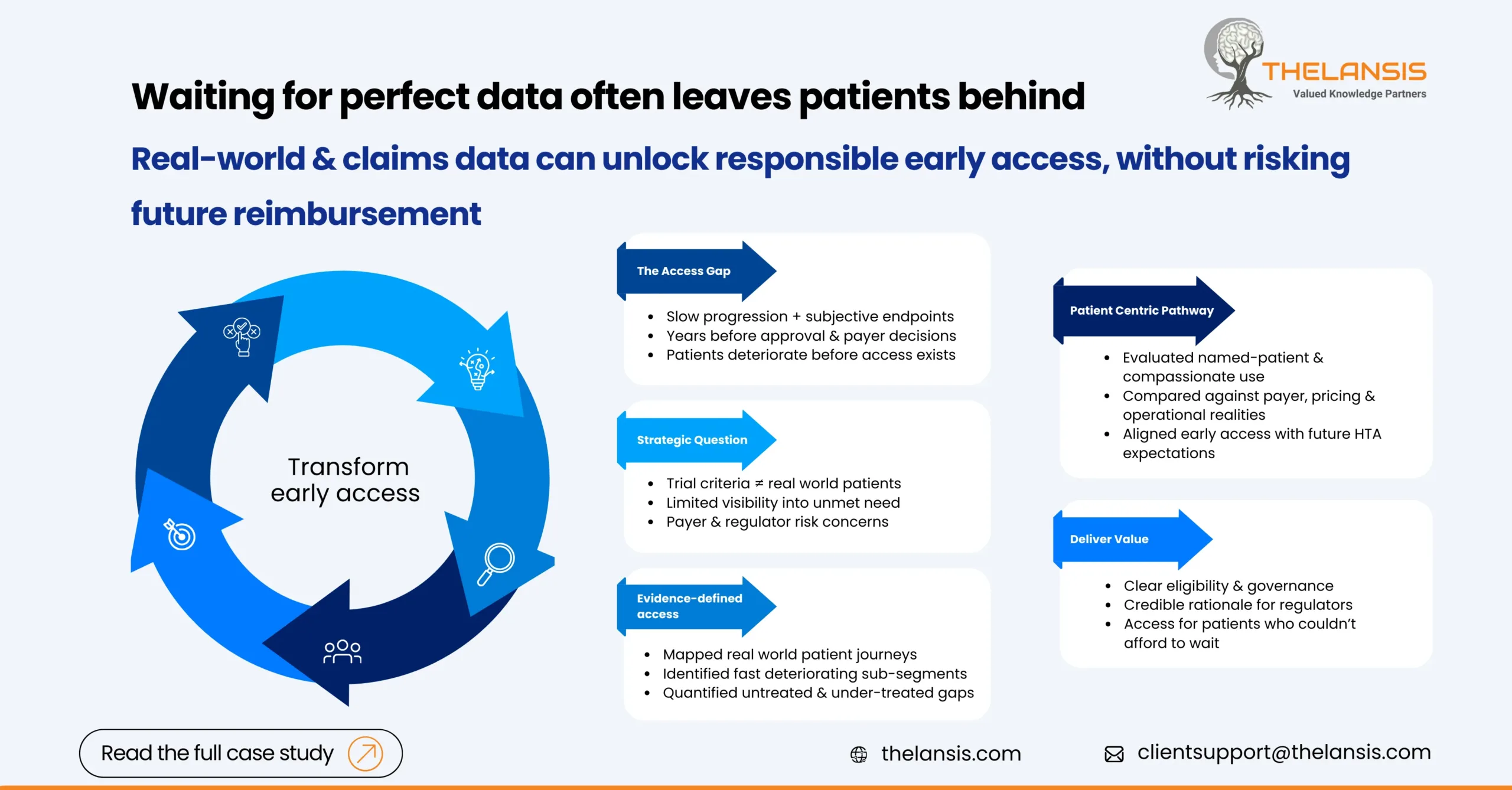
Designing an Early Access Pathway for a CNS Therapy Using Real-World and Claims Insights
Background: A mid-stage biopharma company was advancing a novel CNS therapy into late Phase II, with early clinical signals suggesting meaningful benefit for patients with progressive neurological de ...

The IRA Phase 2: What 2026 Price Negotiations Mean for Innovators, Biosimilars, and Global Launch Strategy
The Inflation Reduction Act (IRA) continues to reshape the U.S. pharmaceutical market. After the first list of 10 Medicare Part D drugs entered negotiations in 2023, the clock is now ticking toward Ph ...

The Collapse & Comeback of Digital Therapeutics: What the Industry Learned and Where It’s Going Next
For a few years, digital therapeutics were treated as inevitable. Prescription apps would scale like software, improve outcomes like medicines, and disrupt chronic care economics along the way. Ventur ...

